Image segmentation for healthcare
Project Brief
Pulmonary acinus represents the gas exchange unit which includes branches of the terminal bronchiole, alveolar ducts, alveolar sacs, alveoli and associated blood vessels. Over the past few decades, many results related to the fluid mechanics characterizing pulmonary acinus of the lungs have been reported. In order to describe a micromechanics in 3D acinar micro-architecture and airflow through it, 3D reconstruction of parenchyma with computational fluid dynamics plays an important role. For the reliable 3D model, precise image segmentation of the stacked 2D images is a necessary pre-step.
Introduction
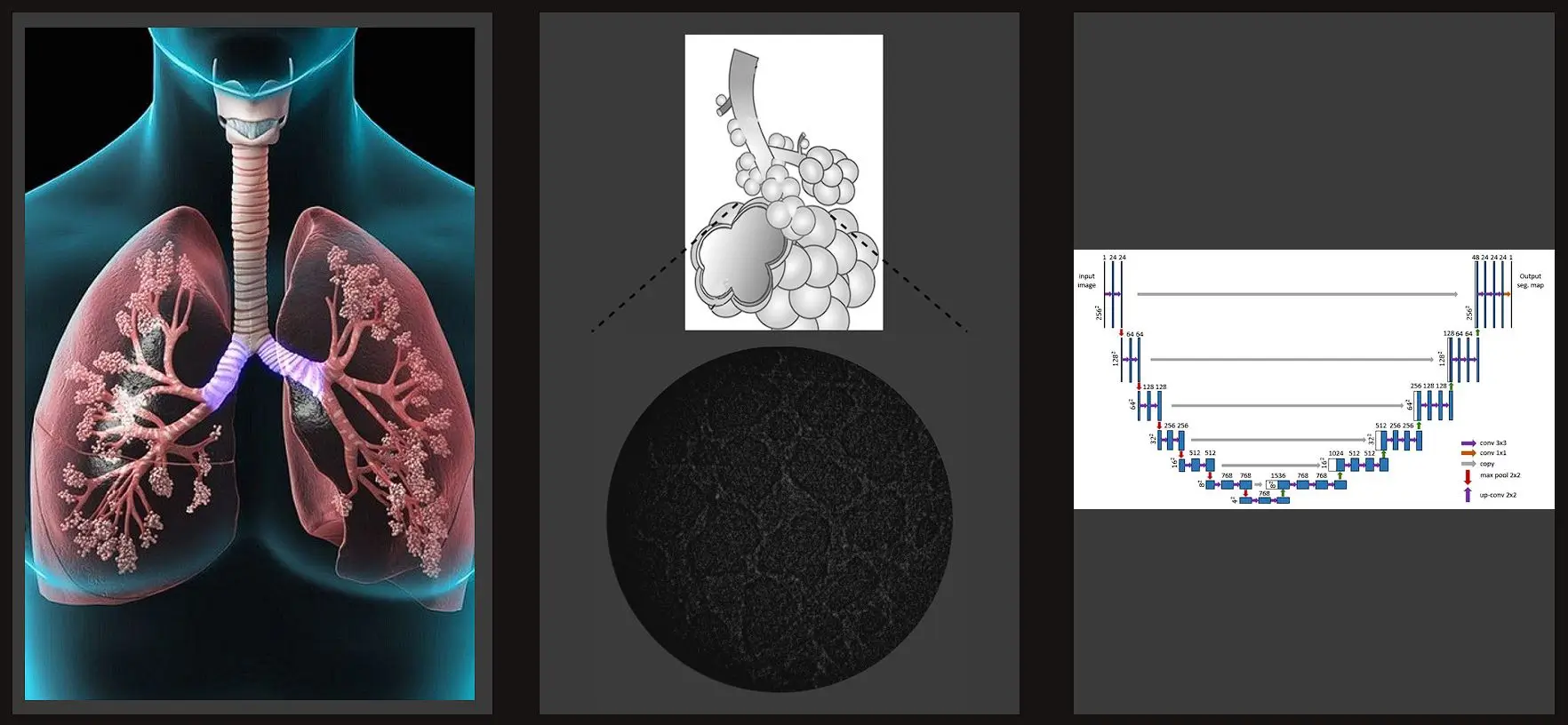
The respiratory tract structure of mammalians plays a vital role in gas exchange function. It is consisted of two areas: conducting/bronchial airways and pulmonary parenchyma. Bronchial airways’ role is to carry the ambient air to parenchyma using its bifurcating network of conduits. Unlike the structure of conducting airways, the structure of airways within parenchyma, called acinar/alveolar ducts, is formed by entrance rings of alveoli followed by opening into a common passageway. The main difference between bronchial and alveolar structure of lungs is their size. This becomes a problem while trying to take the high-resolution images since it cannot be ensured by traditional mCT.
Challange
In the past, the biggest efforts were devoted to the reconstruction of the conducting airways network in three dimensions (3D) from three-dimensional X-ray or CT images. Important role of 3D reconstruction of parenchyma is the study of micromechanics in 3D acinar microarchitecture and airflow through it with the help of computational fluid dynamics. In general, 3D reconstruction of a human pulmonary acinus enables simulating and quantifying regional deposition effects. This task is important in the pulmonary delivery of drugs intended for the topical treatment of respiratory ailments and in situation when lung bioavailability is limited. This task is also important from the perspective of modeling acinar convective airflows and testing their role in determining the fate of inhaled aerosols in the distal regions of the lungs. This way, a patient specific diagnosis and therapy can be conducted. Here we present our new efforts on automatic recognition of alveoli in parenchyma on extracted images as a precondition for 3D reconstruction of acinar air space. The automatic pulmonary acinus (ducts and sacs) segmentation of rat lungs has been done using U-Net based deep convolutional network.
Methodology
U-Net is a convolutional neural network for image segmentation with the most important application being in segmentation of medical images. It is based on encoder-decoder model. Encoder is consisted of convolutional and max-pooling layers which gradually decrease the spatial size of the image and increase the number of channels. After the encoder extracts the features, decoder part symmetrically performs upconvolution and convolution operations. Upconvolution doubles the spatial dimensions of the features, and reduces the number of channels, which corresponds to the way encoder blocks are decreasing image resolution and increasing the depth. In addition, in order to improve quality of the decoder features, and obtain more precise segmentation masks, skip connections are used. They concatenate features from encoder and decoder, therefore decoder which has deep coarse semantic features gets the shallow fine appearance information from the encoder and is able to deliver segmentation masks of the higher quality.
Experimental setup
Stereologically well characterized rat lung samples were imaged using high-resolution synchrotron radiation-based X-ray tomographic microscopy cite{tsuda2008}. As a result, a stack of lung images was generated. Each slice has a size of 1350 x 1350 pixels. These slices had been downsampled to 256 x 256 before they were fed to U-Net. In total, in our dataset we have only 101 labeled (with mask) images, and they belong to different positions in the stack. </p><p>Data augmentation is essential to teach the network the desired invariance and robustness properties, when only a few training samples are available. Because there are not enough images in the training set (only 101 are available), image data augmentation technique is used to artificially expand the size of a training dataset by creating modified versions of images in the dataset. These techniques can create variations of the images that can improve the ability of the fit models to generalize what they have learned to new images. In this way, we can obtain more skillful model.
Experimental results
In the past, the biggest efforts were devoted to the reconstruction of the conducting airways network in three dimensions (3D) from three-dimensional X-ray or CT images. Important role of 3D reconstruction of parenchyma is the study of micromechanics in 3D acinar microarchitecture and airflow through it with the help of computational fluid dynamics. In general, 3D reconstruction of a human pulmonary acinus enables simulating and quantifying regional deposition effects. This task is important in the pulmonary delivery of drugs intended for the topical treatment of respiratory ailments and in situation when lung bioavailability is limited. This task is also important from the perspective of modeling acinar convective airflows and testing their role in determining the fate of inhaled aerosols in the distal regions of the lungs. This way, a patient specific diagnosis and therapy can be conducted. Here we present our new efforts on automatic recognition of alveoli in parenchyma on extracted images as a precondition for 3D reconstruction of acinar air space. The automatic pulmonary acinus (ducts and sacs) segmentation of rat lungs has been done using U-Net based deep convolutional network.
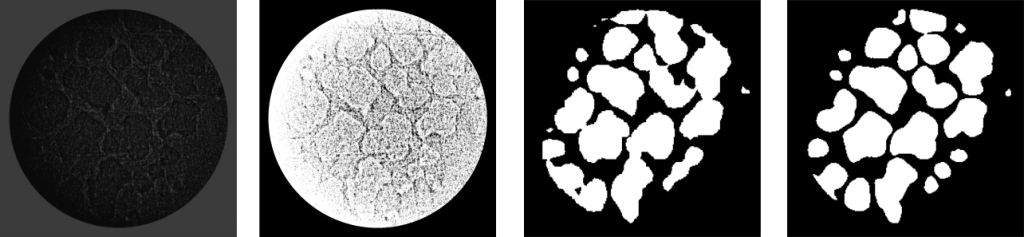
Demo
Team of AI enthusiast with combined professional and academic experience from several industries such as quantitive finance, informatics and bio-informatics, software engineering, optimization , AI and deep learning.
